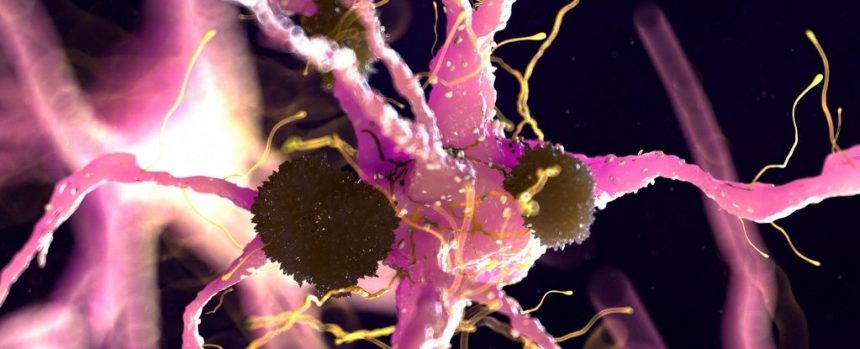
The researchers discovered that a cell surface protein called Aplp1 plays a role in spreading substances that cause Parkinson’s disease from cell to cell in the brain.
Promisingly, FDA-approved cancer drugs target different proteins. Rug 3 – Interact App 1 – It stopped the virus from spreading in mice, suggesting that a potential treatment may already exist.
in New paperAn international team of scientists explains how the two proteins work together to help something harmful. Alpha-synuclein The protein chunks enter the brain cells.
“Now that we know how Aplp1 and Lag3 interact, we have a new way to understand how alpha-synuclein contributes to disease progression in Parkinson’s.” To tell Xiaobao Mao, a neuroscientist at Johns Hopkins University in the US.
“Our findings also suggest that targeting this interaction with drugs may be able to significantly slow the progression of Parkinson’s disease and other neurodegenerative diseases.”
Over 8.5 million Parkinson’s disease patients worldwide The second most common neurodegenerative disease After Alzheimer’s disease.
As it is a progressive movement disorder, it is usually only diagnosed when symptoms such as tremors, stiffness, problems with balance, speech problems, disrupted sleep patterns and mental health issues appear. Currently incurable, the disease means that patients may eventually have difficulty walking and speaking.
The symptoms of Parkinson’s disease are primarily caused by the death or damage of dopamine-producing neurons in the brain. Substantia nigraThis is an area that is involved in fine motor control. Lewy bodiesThese are clumps of abnormal proteins, mainly misfolded alpha-synuclein, that travel between neurons.
Alpha-synuclein normally maintains functional communication between neurons, but when it misfolds and becomes insoluble it causes problems, although it’s difficult to determine whether this is a cause or a symptom of Parkinson’s.
Past research Mouse Lag3 binds to α-synuclein protein “This leads to the spread of Parkinson’s disease pathology to neurons. Deleting Lag3 significantly impedes this process, but does not completely prevent it,” said Dr. Schneider, a professor of neuroscience at the University of California, San Diego, who led the study. “This is a step forward in understanding how misfolded alpha-synuclein can be transported by neurons, and how it can be transported to other neurons.”
“Our previous work demonstrated that Lag3 is not the only cell surface protein that helps neurons take up alpha-synuclein, so in our latest experiments we focused on Aplp1.” To tell Varina Dawson, a neuroscientist at Johns Hopkins University.
The scientists tested genetically modified mice lacking either Aplp1 or Lag3, or both, and found that while Aplp1 and Lag3 each independently help brain cells absorb harmful alpha-synuclein, together they greatly increase that absorption.
When mice lacked both Aplp1 and Lag3, the amount of harmful α-synuclein entering healthy brain cells was reduced by 90 percent — meaning that more harmful clumps of the protein were blocked when both proteins were missing compared with when only one protein was missing.
The researchers gave the drug to normal mice. Nivolumab/Lelatolimabis a melanoma drug that contains an antibody to Lag3, and was found to also block the interaction between Aplp1 and Lag3, almost completely inhibiting the formation of disease-causing α-synuclein clumps in neurons.
“Anti-Lag3 antibodies successfully prevented the further spread of α-synuclein seeds in a mouse model and showed a superior effect to Lag3 depletion due to the close relationship between Aplp1 and Lag3.” To tell Ted Dawson, a neuroscientist at Johns Hopkins University.
The next step will be to test the Lag3 antibody in mouse models of Parkinson’s and Alzheimer’s disease. Research points to Lag3 As a target too.
This study Nature Communications.