health
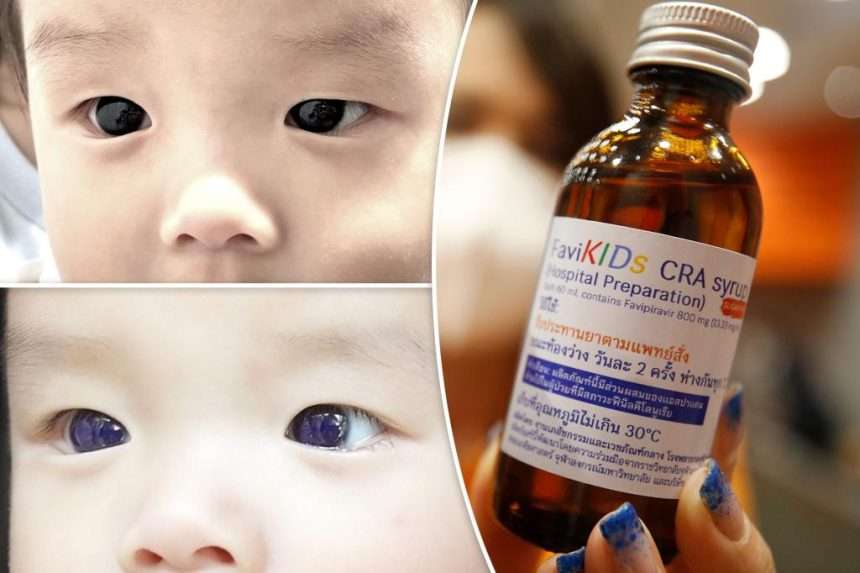
A boy’s eyes temporarily changed from dark brown to bright blue after undergoing treatment for a common coronavirus infection.
The 6-month-old child from Thailand was diagnosed with COVID-19 after suffering from a fever and cough for a day, as detailed in a case published in a medical journal. Frontiers of Pediatrics.
The infant was prescribed the antiviral drug favipiravir. Approved by Ministry of Public Health of Thailand in 2022 For children with mild to moderate symptoms.
Surprisingly, about 18 hours after starting the medication, the child’s mother noticed that his dark brown eyes had turned bright blue.
The concerned mother contacted a medical professional who instructed her to stop treatment immediately.
After about 5 days, the discoloration disappeared and the child’s cornea returned to its original color. Doctors examined the infant and found that the cornea was clear and no longer bluish, and there was no blue pigmentation on the surface of the iris or anterior lens capsule.
Experts say they are not sure why Favipiravir causes the discoloration, but that fluorescence (emission of absorbed light) “may be due to the drug, its metabolites, or additional tablet ingredients such as titanium dioxide or yellow ferric oxide.” “It’s probably something like that,” he speculates.


Previous studies have pointed to a “direct relationship” between favipiravir concentration and fluorescence intensity, particularly in human hair and nails.
In the end, the child did not appear to have suffered any damage to his vision and overcame the symptoms of COVID-19.
The most common side effects of favipiravir include mild hyperuricemia (increased uric acid levels), diarrhea, and neutropenia (reduced levels of white blood cells, neutrophils); Accounts for approximately 20% of side effects.
However, in another recorded case, the drug caused corneal discoloration.
In 2021, a man in India reported. First case of unusual side effects from treatment.




An anonymous 20-year-old man was diagnosed with COVID-19 after two days of no improvement while taking vitamin C, zinc, vitamin A, vitamin D, and ivermectin, and started taking favipiravir. Prescribed.
However, on the second day of Favipiravir treatment, the man noticed that his dark brown eyes had turned bright blue. Doctors advised the man to stop taking the medication, and his eye color returned to normal after just one day.
The report on the infant was released in April 2023, but the exact date of the incident is unknown.


Favipiravir has been approved in Japan, Russia, Ukraine, Uzbekistan, Moldova, Kazakhstan, and received emergency use authorization in Italy in 2020.
U.S. researchers have tested this drug Effective treatment for coronavirus disease (COVID-19) Several studies have tested the drug’s effectiveness in fighting this disease, but it has not been approved by the Food and Drug Administration.
Load more…
{{#isDisplay}}
{{/isDisplay}}{{#isAniviewVideo}}
{{/isAniviewVideo}}{{#isSRVideo}}
{{/isSR video}}