Sexual desire in male mice appears to stem from a single newly identified brain circuit, a new study reveals.
This circuit governs sexual desire and the resulting behavior and reward experience in male mice, and has been suggested to play an important role in forcing mice to reproduce.
That’s interesting, but the US researchers are interested in more than just what motivates sex in mice. They note that this brain circuit is likely ancient, given its apparent importance in encouraging male mice to mate.
The researchers say this basic brain circuitry is unlikely to change significantly over the course of evolution, and there is reason to suspect that it is relatively standard, at least in male mammalian brains.
“We have identified circuits in the male mammalian brain that control sexual cognition, sexual desire, mating behavior and pleasure.” To tell Lead author Nirao Shah is a neurobiologist at Stanford University.
Studying this circuit could provide new insights into mammals as a whole, said Shah and his colleagues, who are working to study the equivalent circuit in female mammals.
Because we are mammals, these insights could also lead to new medicines for humans or shed light on the factors that shape human sexuality.
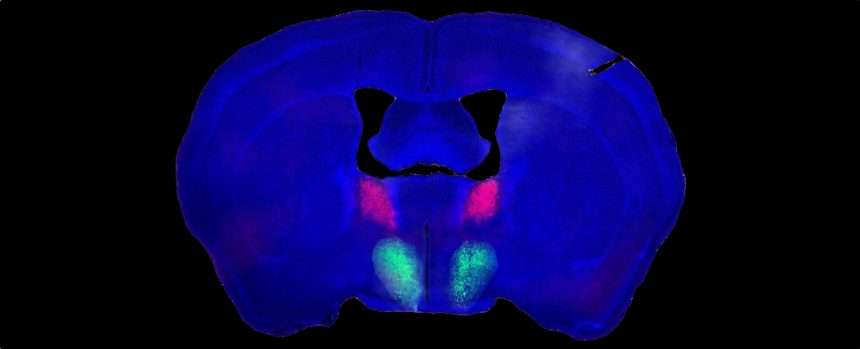
To identify this brain circuit, the researchers studied adult male mice whose behavior and brain activity showed little change due to social influences. Not only were they virgins, they had never even seen a female mouse since they were weaned.
in Previous researchShah and his team were interested in neurons that extend from a part of the amygdala called the bed nucleus. terminal line Link to (BNST) preoptic cortex inside Hypothalamus.
The researchers found that by manipulating these neurons, they could turn on and off the ability of male mice to recognize the gender of unknown mice. New research extends their findings and focuses on neurons.
“We wanted to know exactly which of these neurons were talking to which neurons in the preoptic hypothalamus after recognition had occurred,” Shah says. To tell.
Researchers report that one group of genetically distinct BNST neurons produces a peptide known as substance P, and another unique group of neurons in the preoptic hypothalamus has substance P receptors. ing.
We found that BNST neurons that produce substance P form connections with their receptor counterparts from the preoptic hypothalamus, and that these links serve at least one purpose.
When the researchers used a laser to stimulate specific BNST neurons in the brains of male mice, the corresponding neurons in the preoptic hypothalamus suddenly became active, exhibiting increased activity for about 90 seconds.
Then, after 10 to 15 minutes, the mice begin a series of mating behaviors reliably.
Substance P appears to be key, flowing out of the BNST and gradually stimulating neurons in the preoptic hypothalamus, increasing their activity. When the team played around with this process, they noticed some surprising changes in mouse behavior.
For example, injecting substance P into relevant areas of male mice’s brains significantly improved their ability to mate with willing females.
And when the researchers directly activated preoptic hypothalamic neurons that contain substance P receptors, some male mice even attempted to mate with inanimate objects.
Substance P also caused male mice to ignore the refractory period, a fluctuating period after mating that reduces the willingness and ability of male mammals to mate again.
The mice in this study typically have a five-day refractory period, but stimulating specific neurons in the preoptic hypothalamus allowed the male mice to quickly return to mating, even shortly after termination.
“It took less than a second for them to resume sexual activity,” Shah said. To tell. “This translates into a reduction in the refractory period of more than 400,000 times.”
Suppressing the same neurons made the male mice celibate, Shah added, but they appeared to be otherwise unaffected.
Researchers point out that there are other brain regions important for voluntary movement and pleasure downstream of neurons with substance P receptors, suggesting how this might work, but this It’s not always just the mouse.
“It’s very likely that there is a similar set of neurons in the human hypothalamus that control sexual reward, behavior, and gratification,” Shah says. To tell. “And they’re probably very similar to what we observed in mice.”
The researchers say these findings could aid in the development of drugs to help manage excessive or lack of sexual desire in men, among other possible applications.
“If these centers exist in humans, and now we know where to look, it will be possible to design small molecules that can be used to modulate these circuits.” It should be,” Shah said. To tell.
This research cell.