As a teenager, I worked summers as a ride operator at a small amusement park. Looking back, the whole experience was a lot of fun, but the minimum wage was $3.37 an hour and I was exposed to the changeable New England weather, from freezing rain to heatstroke-inducing tropical sweltering weather. No, I didn’t think it was fun. Like back then.
One of my duties, and one I remember most fondly, was driving bumper cars. Like everything else in the park, the rides were old and worn, and maintenance was a daily chore. To keep the truck’s steel floor from rusting, it had to be brushed with graphite “paint” every morning. It was an incredibly tedious job. Just a little of the oily, silver-black gunk on my hands was there for the rest of the day. And on the first few runs of the day, excited guests were likely not to stuff the material into their shoes before it soaked into the floor, and everyone was sure to step on the seat before sitting in the car. So…well, let’s just say it was easy to spot, especially the one wearing white pants and riding up in a bumper car from behind.
The properties that made graphite ideal for bumper cars (slippery, conductive, tenacious, cheap) are also properties that make it suitable for countless industrial processes. As transportation decarbonization accelerates, its importance is increasing. Graphite is surprisingly useful and fairly common, but it’s not all that easy to extract or purify. So let’s take a look at what it takes to mine and refine graphite.
heat, pressure, insects
The story of graphite begins two billion years ago, when the oceans were teeming with cyanobacteria. These organisms, which had just pulled off the evolutionary trick of figuring out photosynthesis, absorbed nearly unlimited carbon dioxide from the Precambrian atmosphere, turning the upper layers of the ocean into a thick bacterial soup. When they die, mats of carbon-rich remains pile up on the ocean floor, eventually buried under thick layers of sediment, where pressure and heat can begin to work their alchemy.
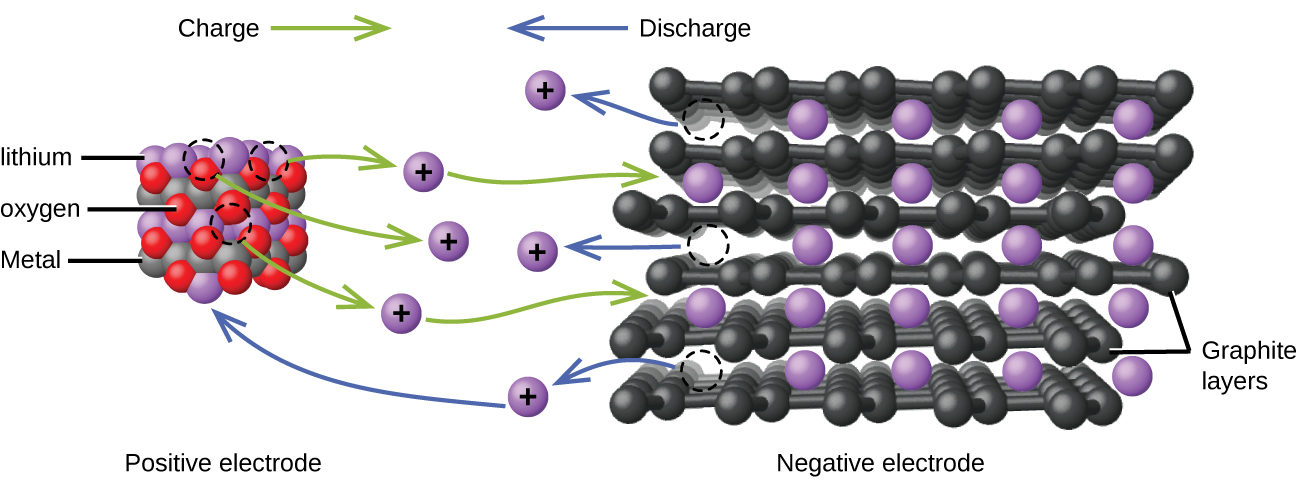
All these dead bacteria are the ultimate source of graphite (not to mention all the other carbon minerals, from coal to oil to diamonds), but exactly what kind of graphite is formed? depends on the geological situation. The most abundant form of graphite is flake graphite. This occurs when dead cyanobacteria form carbon-rich shales and limestones and are forced into high-pressure, high-temperature conditions by tectonic activity. These metamorphic conditions produced rocks with graphite crystals trapped inside. On the other hand, when dead cyanobacteria formed coal deposits, amorphous graphite was formed, and then the coal deposits were swallowed up by the earth’s furnace. The heat and pressure burned away everything but the carbon in the coal, leaving almost completely pure carbon.
Regardless of how graphite is formed, nearly pure carbon crystals give it unique chemical and physical properties. Graphite is a very soft mineral, often as soft as talc. It is formed by stacking multiple one-atom-thick layers of carbon, which gives graphite its fame as a lubricant. In the past, graphite’s lubricity came from the fact that fluids such as air or water are trapped between these monoatomic layers, providing slippage, or simply from the fact that individual sheets are sheared together It was thought that. However, current thinking is that graphite is slippery because adjacent layers are twisted around each other on axes perpendicular to the layer lines. This unlocks the nesting between layers, allowing you to slide them into each other.
In addition to its lubricity, graphite has a vast number of useful properties, some of which have been exploited since ancient times. Neolithic cultures used natural graphite as a colorant in ceramics, and the mineral’s refractory properties (it does not melt until above 3,600°C) have been used for nearly 500 years in high-temperature industrial processes such as crucibles and molds. It has been. Metal casting. Today, graphite has an incredibly wide range of uses, being used in everything from pencil lead to electrodes for smelting aluminum. But graphite’s newest, and perhaps biggest, market is in the production of batteries used in everything from cell phones to electric cars. Approximately 95% of the negative electrode in most lithium batteries is composed of graphite, and it takes 50 to 100 kg of graphite to manufacture a single EV battery.
silver bubbles
There are only a few places in the world where pure graphite deposits exist, and Sri Lanka is the only commercially viable source of this type. And that graphite tends to be reserved for specialized uses. Therefore, like most commercially valuable minerals, mining and refining graphite requires producing enough of the material to justify the labor and expense required to extract it from the rock in which it is embedded. It’s a matter of finding deposits that contain. Graphite mines are typically open-pit mines that typically require extensive equipment such as blasting to quickly extract vast amounts of ore, huge shovels and transport trucks, and crushing plants to reduce graphite-bearing rock to a manageable size. All equipment and processes are in place.
After primary crushing, graphite ore is ground into fine powder. To release graphite from the surrounding rock or gangue, the particle size must be smaller than the graphite flakes within the rock. This is achieved through a series of grinding processes using ball mills and rod mills. Depending on the type of rock and the morphology of the graphite, acids may be used to dissolve the gangue surrounding the graphite. In any case, graphite is most often separated from gangue by some type of flotation process. Because graphite is much less dense than the surrounding rock, it easily forms bubbles that lift the graphite to the top of the flotation tank where it can be skimmed off, while the denser gangue sinks. Flotation also has the added benefit of allowing the graphite to be easily moved to various processes within the plant by pumping it.
The graphite-rich slurry obtained from flotation goes through a series of steps to remove remaining impurities and further concentrate the graphite. These steps include filtration, centrifugation, and separation. Spiral separators are sometimes used. The slurry flows down the helical sluice with lighter particles and water and tends to flow along the outer edge, while the denser gangue particles collect along the inside of the sluice. Once the graphite slurry reaches the desired purity, it is dried in a kiln, sieved to specific sizes, and bagged for shipment.
In 2021, global natural graphite production exceeded 1 billion tons. Almost 80% of this is produced in China, with the remainder mainly produced in Brazil, Turkey and India. There is currently only one operating graphite mine in North America, the Lac des Iles mine in Quebec. Although mines have existed in Montana and Alabama in the past, there are no active graphite mining operations in the United States.
hot coke
However, natural ores are not the only source of graphite. His method of manufacturing artificial graphite has been around since the 1890s, and although it produces graphite of very high purity, it comes at a significant price. Synthetic graphite is made by exposing amorphous carbon to very high temperatures to rearrange the carbon atoms into the proper crystal structure. The selection of raw materials is important in the process of producing high-quality graphite. At some point in its thermal history, carbon must pass through an “intermediate phase,” where most of the work of creating and aligning the basic structural units of carbon crystals has already been completed. The final heat treatment is actually just an annealing process that indexes the monatomic carbon sheets into their final layered shape.

As it turns out, the perfect starting material is petroleum coke or petcoke, a byproduct of oil refining. This carbon-rich material is essentially the low-value material that remains after all the higher distillates, such as diesel and gasoline, are boiled off. Pet coke is essentially a solid mass of long-chain molecules, mostly carbon, but with volatile organics and small amounts of moisture trapped inside, both of which are removed by heating or calcining the coke in a horizontal rotary kiln. It must be removed by: Graphitization takes place in an Acheson furnace, named after Edward Goodrich Acheson, who came across the synthetic graphite process while looking for a way to make synthetic carborundum.
The Acheson furnace is essentially a giant carbon composition resistor packed with calcined pet coke with embedded graphite electrodes at each end of the furnace. Graphitization begins when a large current flows through the graphite electrode, heating the filled pet coke to approximately 2,600℃. This process continues until graphitization is complete. This usually only takes a few days, but cooling the furnace and the graphite inside can take several weeks. This process can be used to produce raw synthetic graphite powder, but is more commonly used to create graphite components such as electrodes for metal refining. In that case, coal tar pitch is mixed with pet coke before firing and acts as a binder giving mechanical strength to the graphite electrode.
Considering the cost of the raw material and the huge amount of energy required to produce it, the cost of synthetic graphite is about twice that of natural graphite. However, there are some applications where it makes sense. The melting electrode mentioned above is a good example because it can be manufactured in its final shape. Synthetic graphite also tends to be suitable for use in composite materials such as carbon fiber.
There is no denying that obtaining graphite, whether natural or synthetic, is a tedious task. Either we burn diesel and drill lots of holes in the ground, or we keep refining oil to run megawatts of electricity through our waste. And since the need for graphite is likely to continue to grow, we hope someone finds a better way.








