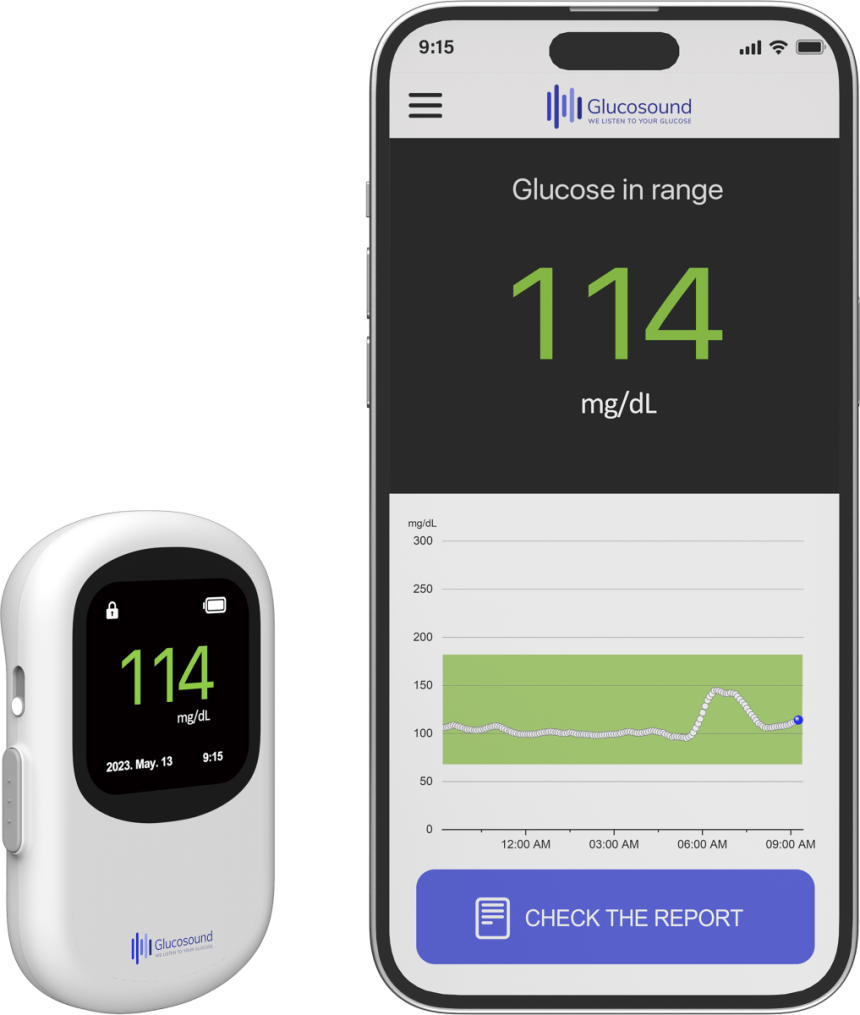
People with diabetes should monitor their blood sugar levels regularly by skin prick. Many glucose monitor makers around the world are pursuing needle-free approaches to make this painful process more manageable. Among them is a South Korean company called HME Arenawhich uses photoacoustics to non-invasively test glucose levels.
HME Square was founded in 2020 by Yunho Khang, who previously worked in semiconductors at Samsung Enterprises for 20 years, and Suh Im, a medical doctor.
Khang tells TechCrunch that the founders wanted to develop a device to address this common health issue and decided to build a needle-less glucose monitor. Diabetes affects more than half a billion people worldwide, and is expected to do so To reach 1.3 billion by 2050.
HME Square, a member of this year’s Battlefield 200 at TechCrunch Disrupt, claims its preliminary study shows better accuracy than continuous glucose monitors (CGM) on the market. A preliminary study shared with investors, based on the Mean Absolute Relative Difference (MARD) and the Clark Error Grid (CEG), a tool used to evaluate the accuracy of glucose monitors, showed that the current average absolute relative difference is 7%. (The lower the MARD, the greater the accuracy.) CGMs, such as those from Abbott, Dexcom and Medtronic, show between 8.7% and 9.2%.
Khang said the company’s device uses a MEMS sensor and photoacoustics using deep learning to measure blood sugar. Optical acoustics It is a non-invasive sensing technique that involves shining light on a substance (such as blood) and reading the ultrasound waves it emits.
Kang points out that this technology could also be used for broader applications in various biological materials in the body. For example, it can monitor ketones, glycated hemoglobin (HbA1c), and cholesterol in the second phase, and bilirubin and creatinine in the third phase.
Kang said HME Square is in the research and development phase now, and it will take a few more years to bring the device to market in South Korea and the United States, with a goal of 2025. The startup plans to implement Good Manufacturing Practices (GMP) In Korea next year. GMP is a set of standards that ensure that pharmaceutical products are produced consistently. Khang adds that after obtaining GMP approval, he will begin clinical trials in the country, which will take about a year.
Unlike intrusive measures, such as blood sampling or continuous glucose monitoring, which can easily establish a reference point because samples are collected directly, Khang explains that with non-intrusive glucose monitoring, it is not easy to obtain accurate results or data.
“For example, visual method technology Used by Apple It obtains a lot of data, but because it cannot directly compare signals from different wavelengths, it is difficult to improve the measurement accuracy. “We are focused on solving this problem, and are currently making significant progress.”
Khang claims that their photoacoustic method is superior because the photoacoustic ultrasound signals are made of glucose absorbed energy from the laser and contain more information, though says a recent report“Among non-invasive glucose monitoring techniques, optical methods give the best measurements. Optical techniques such as near-infrared, mid-infrared or Raman spectroscopy have great selectivity for glucose sensing due to the complexity of blood/tissue properties.
The company will use a direct-to-consumer business model, selling products to people with diabetes and people using invasive procedures such as CGM. The company is also considering adding subscription models once it’s commercialized, Khang says, adding that its device will likely cost around $1,000 for two years of use.
Since its inception, the company has raised more than $3 million from investors, including Posttech Holdings. HME Square is currently raising $2 million for research and development and hiring additional employees.