Lecanemab received FDA approval in January. Phase 3 trial showed slowing cognitive decline For patients with early stage Alzheimer’s disease.
We know lecanemab removes amyloid beta buildup, but it wasn’t clear how – But new research from New York’s Rockefeller University sheds some light on that. This could pave the way for new treatments for Alzheimer’s and other diseases.
“We believe we have discovered a mechanism that is one of the reasons why lecanemab works.” To tell Neurobiologist Erin Norris.
This drug appears to inhibit the plasma contact system. An interaction between proteins in the blood that helps promote clotting and inflammation. Although this system helps repair tissue damage, it can cause harm if sensitive areas such as brain tissue become overstimulated, putting us at risk for conditions such as Alzheimer’s disease.
“If you block the contact system, you will develop less Alzheimer’s disease.” To tell Neurobiologist Sidney Strickland.
Brain disorders, particularly neurodegenerative diseases such as Alzheimer’s disease, are among the most difficult medical problems to solve. Alzheimer’s disease affects millions of people worldwide and there is currently no cure.
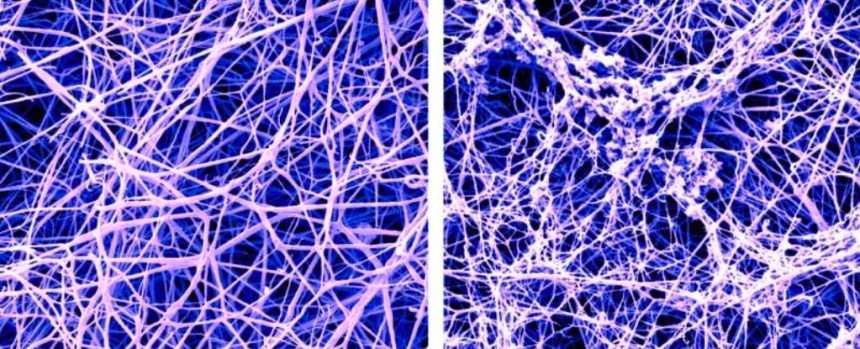
two important proteins, Tau and amyloid beta (Aβ) aggregates into tangles and plaques in Alzheimer’s disease, leading to brain cell death and brain volume loss.
Aβ plaques are formed from different types of aggregates, of which protofibrils are thought to be the most toxic. It is these forms of plaque that lecanemab targets.
Until recently, treatments aimed at removing plaque from patients’ brains have not been able to significantly slow cognitive decline.
However, patients who took recanumab During the 18 month trial period Compared to people who took a placebo, there was a statistically significant reduction in amyloid beta plaques in the brain and a 27 percent slower decline in cognitive function.
Unfortunately, as is often the case, this treatment is not without negative effects. This includes an increased risk of mild to moderate bleeding and swelling in the brain, called cerebral hemorrhage. Amyloid-related imaging abnormalities (Aria). However, the risks are relatively improved when compared with similar plaque-destructive treatments.
“It is noteworthy that lecanemab causes less ARIA (10%) than other anti-Aβ antibodies such as aducanumab (35%), gantenerumab (30%), and donanemab (27%),” the research team said. . written in their paper.
Lecanemab’s low ARIA rate piqued the interest of Norris, Strickland, and colleagues, who wanted to know why it was so effective.
After extensive analysis of plasma from eight donors without Alzheimer’s disease, researchers discovered that protofibrils are the only form of Aβ that is just the right size to activate the plasma contact system.
Norris: “It’s really amazing.” To tell“The form of Aβ that specifically activates the contact system is the same form of Aβ targeted by lecanemab.”
Activation of this contact system leads to overproduction of a peptide called . bradykininThis can dilate blood vessels and cause ARIA side effects, which are common with other anti-Aβ therapies.
Lecanemab appears to act by reducing Aβ accumulation and preventing protofibrils from activating the contact system, resulting in a decrease in bradykinin production.
The research team developed an antibody called 3E8 target circulating plasma proteins You will get similar results. They believe their findings mean that 3E8, alone or in combination with drugs like lecanemab, has potential in the treatment of Alzheimer’s disease, and perhaps could have broader effects. Masu.
There’s a long way to go, but depending on how well it performs in clinical trials, researchers believe 3E8 may have the potential to treat other diseases as well.
“Dysregulation of the contact system has been implicated in the spread of coronavirus, sickle cell anemia, hereditary angioedema, inflammatory bowel disease, sepsis, lupus, arthritis, and even cancer metastasis,” says Norris. he said. To tell.
The study was published in the journal Proceedings of the National Academy of Sciences.