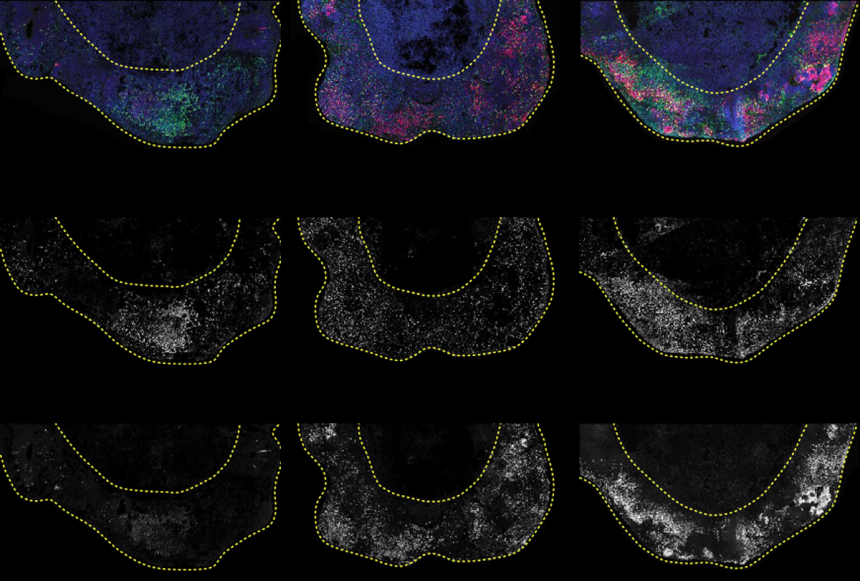
Mutation combinations: After 40 days of growth, cerebral organoids show interneuron progenitors (green, top row, bright dots, middle row) and oligodendrocyte progenitors (red, top row, bright dots, bottom row). ARID1B mutations (middle and right columns) lead to more cells becoming early oligodendrocyte precursors compared to control organoids (left column).
a New genetic screening technology reveals how dozens of mutations associated with autism alter the fate of brain cells during development in organoids.
“We wanted to find stages of brain development that are particularly susceptible to autism-related genetic mutations,” says the co-principal investigator. Jurgen KnoblichProfessor of Synthetic Biology at the Medical University of Vienna, Austria.
This approach builds on the methods of Knoblich’s research group. developed In 2020, we aim to screen genes associated with microcephaly for their ability to inhibit cell proliferation. In that study, the team used CRISPR to create a series of genetic mutations in human stem cells, grew each stem cell into an organoid, and tracked the mutations that accumulated in each cell using a unique genetic barcode. .
In their new book published in September, Nature, the team used a similar approach to generate human organoids with mutations in 36 genes strongly associated with autism. Each cell in the mosaic organoids contained a single mutation.
“This is the first time I’ve seen many genes tested at once in the same model,” he says. Flora Vaccarino, a professor of neuroscience at Yale University, was not involved in the study. “It’s really amazing.”
After the organoids had grown for four months and accumulated thousands of cells with each mutation, Knoblich and colleagues used single-cell RNA sequencing to measure the abundance of individual cell types.
COral organoids typically grow a mixture of neurons and glia arranged in layers similar to those found in the human cerebral cortex. The researchers found that mutations in 24 of the 36 genes altered the ratio of excitatory neurons to inhibitory interneurons. Approximately one-third of the mutations reduced the number of intermediate progenitor cells. Intermediate progenitor cells give rise to excitatory neurons throughout cortical layers and are thought to be important in integrating information across brain regions.
“This problem of imbalance seems to be a common denominator in the development of autism spectrum disorders,” Vaccarino says.
In particular, mutations in certain genes, ARID1B, produced one of the “strongest and most interesting” results in organoids, Knoblich said. The ARID1B mutation prompted more cells than normal to become oligodendrocyte precursors. produce electrical insulation It exists around neurons and is thought to be associated with autism.
The study showed that cerebral organoids derived from two people with ARID1B mutations both showed an abnormal increase in oligodendrocyte progenitor cells, similar to the CRISPR engineered organoids. “We went from genetic screening to actually recruiting patients with the mutation, and we’re very proud that we were able to see this through,” Knoblich said.
Replicating the ARID1B findings in human-derived organoids is the first step toward validating that the findings apply to the human brain, Knoblich said. Vaccarinio added that future studies should continue to examine more people with various other genetic mutations associated with autism.
The 36 genes that Knoblich’s team focused on are all involved in transcriptional regulation, which controls how cells respond to intracellular and extracellular signals. However, many other autism-related genes are involved in forming and maintaining connections between neurons.
Knoblich says the new approach can tackle not only this set of genes, but many others. “This is a very versatile technology. Any list of genes can be built into the system and used for any organoid.”